Abstract
Introduction: Acalabrutinib (Calquence ®), a selective Bruton tyrosine kinase (BTK) inhibitor, is approved for the treatment of mantle cell lymphoma (relapsed/refractory) and chronic lymphocytic leukemia. Patients with hematologic malignancies may require acid-reducing agents (including proton pump inhibitors [PPIs]) for the treatment of gastroesophageal reflux or peptic ulcer disease. The solubility of acalabrutinib is reduced with increasing pH; concomitant administration of acalabrutinib capsules with PPIs reduces acalabrutinib exposure and is currently not recommended. Additionally, many cancer patients are unable to swallow capsules and require alternative methods to deliver acalabrutinib, such as a suspension administered orally or via a nasogastric (NG) tube. To enable the use of acalabrutinib in patients who require co-treatment with PPIs and/or are unable to swallow capsules, a new maleate salt of acalabrutinib, formulated as an immediate-release film-coated tablet (acalabrutinib maleate tablet [AMT]), has been developed which shows fast and complete in vitro release at all physiologic pH. We evaluated the pharmacokinetics (PK), pharmacodynamics (PD), safety, and tolerability of AMT administered orally or via NG tube in the presence or absence of a PPI. In addition, the effect of food on AMT was evaluated to confirm the absence of a clinically relevant impact, consistent with acalabrutinib capsules.
Methods: Three Phase 1, open-label, single-dose, cross-over studies were conducted in healthy subjects to establish PK similarity (bioequivalence) between 100-mg AMT and 100-mg acalabrutinib capsules (N=66); evaluate PPI effect by comparing PK of 100-mg AMT administered in the presence vs absence of rabeprazole (PPI; N=14); evaluate food effect by comparing PK of 100-mg AMT administered with a high-fat diet vs fasted (N=16); and assess PK following administration of 100-mg acalabrutinib maleate suspension (in 15 mL water) delivered via NG tube, in the presence vs absence of rabeprazole (N=20). PD was assessed by measuring BTK target occupancy (BTK-TO) in peripheral blood mononuclear cells across all treatment arms and studies.
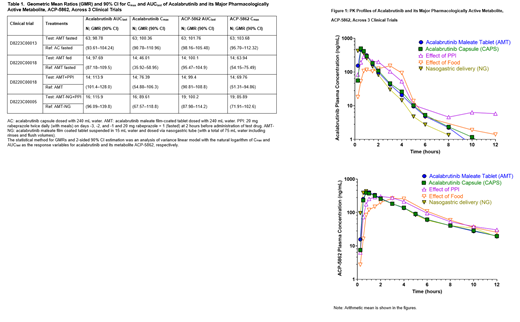
Results: Exposure geometric mean ratios and 90% confidence intervals (CIs) are shown in Table 1 with the PK profiles shown in Figure 1. Systemic exposures (C max and AUC) of acalabrutinib and its major pharmacologically active metabolite, ACP-5862, between AMT and acalabrutinib capsules were bioequivalent (<5% difference in geometric mean exposures, with the 90% CI contained entirely within the pre-defined range of 80.00% and 125.00%). No clinically relevant difference in acalabrutinib/ACP-5862 exposures was observed following administration of AMT with and without PPI; C max was lower (≤~30% difference) and AUC higher (≤~16% difference) with similar BTK-TO (≥95%) across treatment arms. Additionally, no clinically relevant impact of food on acalabrutinib/ACP-5862 exposures was observed; C max was lower (≤~54% difference), with no impact on AUC (≤~3% difference) or BTK-TO (≥95% across treatment arms). Acalabrutinib/ACP-5862 exposures were comparable (≤10% difference) between 100-mg acalabrutinib maleate NG suspension and 100-mg acalabrutinib capsules. In addition, exposures were comparable (≤16% difference) following co-administration of acalabrutinib maleate NG suspension with and without PPI. Overall, the BTK-TO was comparable (≥95%) across all treatment arms. The new AMT formulation showed a well-tolerated safety profile with the majority of observed adverse events (AEs) mild in intensity and no serious AEs reported. No new safety concerns were observed for the AMT.
Conclusions: Acalabrutinib maleate, administered as a tablet or suspension, is safe and well tolerated. Based on the PK (and associated variability), BTK-TO, and established exposure-efficacy/safety relationship, AMT clinical effect is expected to be comparable to acalabrutinib capsules at the approved 100-mg BID dosing, regardless of use of PPIs and ingestion of food. Additionally, AMT improves swallowing ability given the film coating and a 50% reduced volume compared with the capsule, and can be easily suspended in a small amount of water to allow dosing in patients unable to swallow tablets.
Sharma: AstraZeneca: Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Pepin: AstraZeneca: Current Employment. Burri: AstraZeneca: Current Employment, Divested equity in a private or publicly-traded company in the past 24 months. Zheng: AstraZeneca: Current Employment; Kite Pharma, a Group of Gilead: Ended employment in the past 24 months; Gilead Science Inc., AstraZeneca: Current equity holder in publicly-traded company; Gilead Science Inc.: Divested equity in a private or publicly-traded company in the past 24 months. Kuptsova-Clarkson: AstraZeneca: Current Employment, Current equity holder in publicly-traded company; AbbVie: Current holder of individual stocks in a privately-held company. de Jong: Acerta Pharma B.V. (A Member of the AstraZeneca Group): Current Employment. Yu: AstraZeneca: Current Employment; EMD Serono Research Institute: Ended employment in the past 24 months; AstraZeneca, Johnson and Johnson, AbbVie, Abbott: Current equity holder in publicly-traded company; Merck KGaA: Divested equity in a private or publicly-traded company in the past 24 months. MacArthur: AstraZeneca: Current Employment, Current holder of individual stocks in a privately-held company, Current holder of stock options in a privately-held company. Majewski: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Ware: AstraZeneca: Current equity holder in publicly-traded company; Denali (DNLI) Therapeutics: Current equity holder in publicly-traded company. Mann: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Ramies: AstraZeneca: Consultancy. Munugalavadla: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Sheridan: AstraZeneca: Current Employment, Current equity holder in publicly-traded company. Tomkinson: AstraZeneca: Current Employment, Current equity holder in publicly-traded company.
New Formulation

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal